
As Chlorine already had seven electrons in its valence shell, the octet of all the Chlorine atoms is now complete. Step 4: In the above structure, every single bond represents a shared pair of electrons. This helps us estimate the number of electrons further needed to complete the octet of all the involved atoms. Step 3: Now, we will connect all the other atoms of this molecule to the central atom, with the help of single bonds. In the case of XeCl 4, Xenon is the central atom. Mostly, the least electronegative atom of a molecule is chosen as the central atom. To draw the Lewis structure, the central atom is considered the base to which all other atoms of the molecule are attached. Step 2: In the next step, we will have to choose the central atom for this molecule. In the case of the XeCl 4 molecule, Xenon being a member of group 18 of the periodic table carries eight valence electrons while Chlorine being a member of group 17 carries 7 electrons in its valence shell.Ĭalculating the total number of valence electrons: Step 1: First of all, we will calculate the total number of valence electrons present in this molecule. In this section, we will try to draw the Lewis structure of the XeCl 4 molecule, step by step: it is capable of accommodating more than eight electrons in its valence shell. 12.ĭue to the presence of empty 5d-orbitals Xenon is able to expand its octet i.e. However, Xenon that already had a complete octet now carries more than eight electrons in its valence shell i.e. 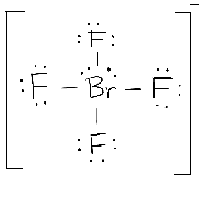
The Lewis structure of XeCl4 is as shown below:Īs per the above Lewis structure for the XeCl 4 molecule, all the Chlorine atoms that initially carried seven valence electrons form a single bond with Xenon and now have eight electrons. These are the pictorial representation of the valence electrons around an atom.Īccording to these structures, the symbol of the element represents the nucleus as well as the electrons in the inner shells, while the valence electrons are represented by using dots. Lewis introduced Lewis structures in 1916. Hydrogen also follows the standards of its nearest noble gas (helium) and becomes stable after acquiring two electrons. Helium is a noble gas that is stable with two electrons in its valence shell. The only exceptions to this rule are Helium and Hydrogen. It is owing to this tendency of atoms that they participate in bond formation. Usually, the atoms try to acquire the electronic configuration of their nearest noble gas to become stable. This was proposed by Kossel and Lewis in 1916. The atoms of all other elements of the periodic table also tend to become stable by having eight electrons in their outermost energy level. They have a valence shell containing eight electrons which are responsible for their stability. The noble gases are the least reactive elements of the periodic table. Hence, it contains 36 electrons in its valence shell. In the case of XeCl 4 Xenon carries 8 electrons in its outermost shell while each of the Chlorine atoms has 7 valence electrons. They can be transferred or shared with other atoms during the process of chemical bonding. (g) Six electron groups give an octahedral electron geometry, while four bonding groups and two lone pairs give a square planar molecular geometry.The electrons situated in the outermost shell, or energy level, of an atom, are known as valence electrons. (f) Six electron groups give an octahedral electron geometry, while five bonding groups and one lone pair give a square pyramidal molecular geometry. (e) Five electron groups gives a trigonal bipyramidal electron geometry, while two bonding groups and three lone pairs give a linear geometry. 
(d) Five electron groups give a trigonal bipyramidal electron geometry, while three bonding groups and two lone pairs give a T-shaped molecular geometry. (c) Five electron groups give a trigonal bipyramidal electron geometry, while four bonding groups and one pair give a seesaw molecular geometry. 
(b) Four electron groups give a tetrahedral electron geometry, while two bonding groups and two lone pairs give a bent molecular geometry. (a) Four electron groups give a tetrahedral electron geometry, while three bonding groups and one lone pair give a trigonal pyramidal molecular geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
